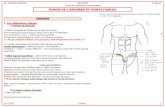
Sels de Bases Faibles Monovalentes Et d'Acide Fort
-
Upload
anonymous-fikonlap -
Category
Documents
-
view
216 -
download
0
Transcript of Sels de Bases Faibles Monovalentes Et d'Acide Fort
-
7/25/2019 Sels de Bases Faibles Monovalentes Et d'Acide Fort
1/1
de bases faibles monovalentes et d'acide fort
//C|/Users/Hp/Desktop/chimie%20des%20solutions/Sels%20de%20bases%20faibles%20monovalentes%20et%20d'acide%20fort.htm[21/02/2016 16:52:
Acides/Bases
Table des
matires
1. INTRODUCTION2. Dissociation de l'eau
et pH3. Concept d'acide et de
base4. Acides polyprotiques
et bases polyvalentes5. Hydrolyse des sels
1. Sels d'acidesfaiblesmonoprotiqueset de base forte
2. Sels de basesfaiblesmonovalenteset d'acide fort
3. Sels d'acidesfaiblesbiprotiques
4. Sels d'acidesfaiblestriprotiques
5. Exercices6. Rsum du calcul du
pH
Sels de bases faibles monovalentes et d'acide fort
Ces sels se dissocient totalement suivant l'expression gnrale :
BHXBH++ X- (22)
Pour les chlorures, X-sera l'ion Cl -prsent la concentration Cselde la solution. La
nature du cation BH+dpend de la base faible dont drive le sel : pour les sels drivs
l'ammoniac, BH+est l'ion ammonium NH4+.
Les ions B+captent une petite quantit d'ions OH-provenant de l'quilibre de dissocia
de l'eau ((1) p.B) ce qui dplace cet quilibre vers la droite. Il se forme donc des ions Hle milieu devient lgrement acide. On peut reprsenter cette raction d'hydrolyse du separ l'quation globale :
BHX + H2OB + X-+ H3O
+ (23)
La relation (13b) p.C5 caractrisant l'quilibre (12) p.C5 peut tre rsolue en considranque [BH+] Cselet [B] [H
+], ce qui fournit :
Ka= = ([B] [H+] / [BH+] [H+]2/ Csel
soit, en passant au logarithme
pH = 1/2 (pKa- log Csel)
(24)
Exemple : pour une solution 0,1 mol/L de chlorure d'ammonium (pKa= 9,25 pour
l'ammoniaque) pH = 1/2 (9,25 - log 0,1) = 5,12.
uilt With Wimba Create
http://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_01.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_02.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_02.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_05.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_05.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_15.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_15.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_16.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_21.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_22.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_22.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_22.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_22.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_21.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_20.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_16.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_15.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_15.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_05.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_05.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_02.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_02.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_01.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_17.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/index.htmhttp://c%7C/Users/Hp/Desktop/chimie%20des%20solutions/page_19.htm